Editas (EDIT) Focuses on Developing Gene-Editing Eye Drug
Editas Medicine, Inc. EDIT has made rapid progress in the development of its lead pipeline candidate, EDIT-101, which employs CRISPR gene editing, to treat Leber congenital amaurosis type 10 (LCA10) — a rare genetic illness that causes blindness.
The company is evaluating EDIT-101 in the phase I/II BRILLIANCE study. Initial data from the same were announced in September 2021. Early observations from subjects treated in the mid-dose cohort of the study demonstrated clinical evidence of gene editing led by visual improvements.
Editas plans to complete dosing in the adult high-dose and pediatric mid-dose cohorts in the first half of 2022.
Shares of Editas have plunged 72.2% in the past year compared with the industry’s decrease of 35.1%.
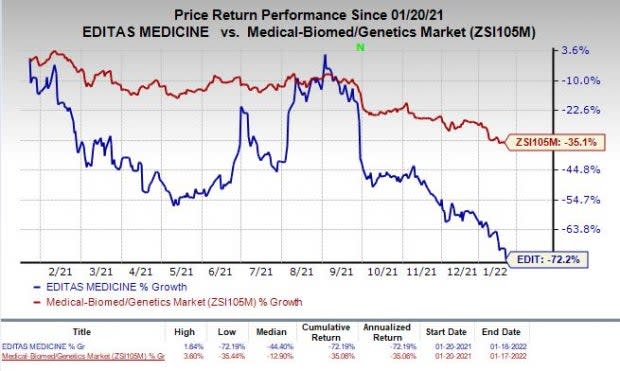
Image Source: Zacks Investment Research
Editas’ other pipeline candidates are also progressing well. The company is evaluating the safety and efficacy of EDIT-301 for treating sickle cell disease (“SCD”). It is currently enrolling participants in the phase I/II RUBY study and expects to begin dosing in the first half of 2022.
In December 2021, the FDA cleared the investigational new drug application for EDIT-301 to treat transfusion-dependent beta thalassemia (“TDT”). The company plans to begin clinical studies on EDIT-301 in TDT patients later in the ongoing year.
Editas currently has no approved product in its portfolio. Therefore, successful development of EDIT-101, along with other pipeline candidates, remains in key focus for the company. Moreover, Editas’ pipeline candidates are in early-stage development and years away from commercialization. Hence, any regulatory setback for the pipeline candidates will be a significant impediment for the company.
Meanwhile, stiff competition remains a headwind as several other companies, including CRISPR Therapeutics CRSP and Intellia Therapeutics NTLA, are engaged in developing candidates to address different indications using CRISPR/Cas9 gene-editing technology.
CRISPR Therapeutics’ lead candidate, CTX001, a CRISPR gene-edited therapy, is being developed for treating SCD and TDT. CRISPR Therapeutics expects to file regulatory submission for CTX001 in both indications by 2022-end.
Intellia Therapeutics is developing NTLA-2001, its lead program in vivo genome editing candidate, as a curative treatment for transthyretin amyloidosis. NTLA-2001 was the first candidate in Intellia Therapeutics’ pipeline to enter clinical development.
Editas Medicine, Inc. Price and Consensus
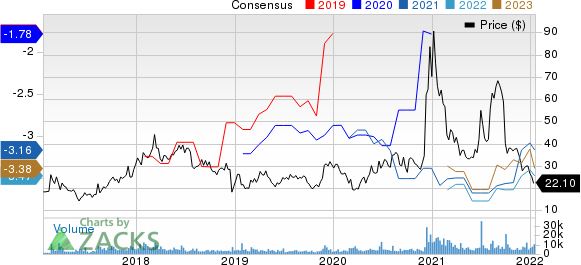
Editas Medicine, Inc. price-consensus-chart | Editas Medicine, Inc. Quote
Zacks Rank & Stock to Consider
Editas currently carries a Zacks Rank #3 (Hold). A top-ranked stock in the biotech sector is AnaptysBio, Inc. ANAB, which has a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
AnaptysBio’s loss per share estimates have narrowed 0.3% for 2022, over the past 60 days. The stock has rallied 25.3% in the past year.
AnaptysBio’s earnings have surpassed estimates in three of the trailing four quarters and missed the same on the other occasion.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Editas Medicine, Inc. (EDIT) : Free Stock Analysis Report
Intellia Therapeutics, Inc. (NTLA) : Free Stock Analysis Report
CRISPR Therapeutics AG (CRSP) : Free Stock Analysis Report
AnaptysBio, Inc. (ANAB) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 