Celldex (CLDX) Initiates Subcutaneous Skin Disorder Drug Study
Celldex Therapeutics, Inc. CLDX announced that it has initiated dosing in the first cohort of a phase I study evaluating a subcutaneous formulation of its monoclonal antibody candidate, CDX-0159. The company intends to evaluate the subcutaneous CDX-0159 as a potential treatment for a skin disorder, chronic urticarias, in a phase II study, which is likely to start in the first half of 2022.
The early-stage has been designed with four cohorts to evaluate four doses — 50 mg, 150 mg, 300 mg, and 600 mg — of the subcutaneous CDX-0159 in healthy volunteers primarily for safety and tolerability. The study’s secondary endpoints include pharmacokinetics, pharmacodynamics, and immunogenicity.
We note that Celldex is already evaluating an intravenous formulation of CDX-0159 in multiple early-stage studies as a potential treatment of skin disorders — chronic spontaneous and chronic inducible urticarias. The company also plans to start a phase I study later this year to evaluate the intravenous CDX-0159 for treating prurigo nodularis, another skin disorder.
A subcutaneous formulation of any drug provides a better and easy administration alternative compared to the intravenous formulation. Several subcutaneous drugs are available with a self-administration option while intravenous drugs are usually physician-administered and require a visit to the hospital or a physician. A subcutaneous formulation of CDX-0159 is likely to be garner more demand than the intravenous one due to its easy and at-home administration potential.
Shares of Celldex have rallied 208.6% so far this year compared with the industry’s increase of 0.8%.
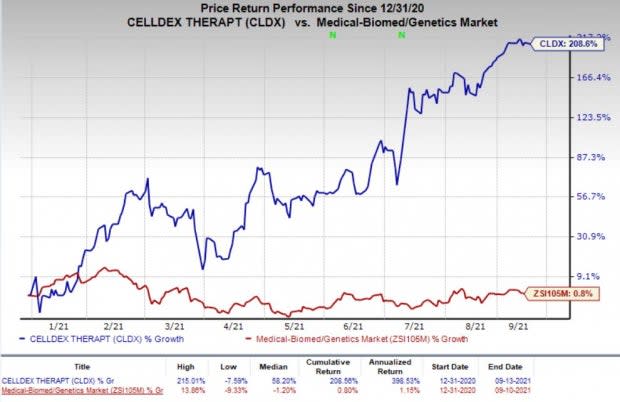
Image Source: Zacks Investment Research
Interim data, announced in July, from a phase Ib evaluating the intravenous formulation of CDX-0159 in patients with chronic inducible urticaria demonstrated a statistically significant response rate. The patients showed sustained durability of the clinical response. The antibody candidate also achieved a complete response in three CIU patients who were previously treated with Novartis NVS/Roche’s RHHBY urticaria drug, Xolair (omalizumab). Two among these patients were refractory to Xolair treatment.
Apart from CDX-0159, Celldex is developing a CD40 human agonist antibody CDX-1140. The candidate is being developed as a potential treatment of solid tumors and B cell lymphomas as a monotherapy or in combination with Celldex’s CDX-301 or Merck’s MRK Keytruda.
The company initiated an early-stage study earlier this year to evaluate its new bispecific product candidate, CDX-527 for solid tumors.
Celldex Therapeutics, Inc. Price
Celldex Therapeutics, Inc. price | Celldex Therapeutics, Inc. Quote
Zacks Rank
Celldex currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Roche Holding AG (RHHBY) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
Celldex Therapeutics, Inc. (CLDX): Free Stock Analysis Report
To read this article on Zacks.com click here.

 Yahoo Finance
Yahoo Finance 