Myriad Genetics, Inc. (MYGN)
After hours:
| Previous Close | 18.64 |
| Open | 18.57 |
| Bid | 18.46 x 200 |
| Ask | 18.52 x 200 |
| Day's Range | 18.22 - 18.91 |
| 52 Week Range | 13.82 - 24.21 |
| Volume | |
| Avg. Volume | 620,435 |
| Market Cap | 1.675B |
| Beta (5Y Monthly) | 1.98 |
| PE Ratio (TTM) | N/A |
| EPS (TTM) | -3.18 |
| Earnings Date | May 01, 2024 - May 06, 2024 |
| Forward Dividend & Yield | N/A (N/A) |
| Ex-Dividend Date | N/A |
| 1y Target Est | 23.91 |
 GlobeNewswire
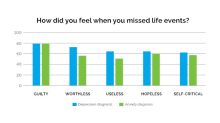
GlobeNewswireNearly Half of Americans Say They’ve Lost Time in Their Lives Due to Poor Mental Health, According to the GeneSight Mental Health Monitor
80% of people diagnosed with anxiety, depression report losing years or decades of timeSALT LAKE CITY, April 17, 2024 (GLOBE NEWSWIRE) -- An astonishing 44% of Americans feel they have lost time in their lives due to poor mental health. That number jumps to nearly 80% for those diagnosed with anxiety and/or depression, according to the latest GeneSight Mental Health Monitor, a nationwide survey from Myriad Genetics, Inc. (NASDAQ:MYGN), a leader in genetic testing and precision medicine. Of those
 GlobeNewswire
GlobeNewswireNew Myriad Genetics Study Published in Prenatal Diagnosis Shows High Positive Predictive Value for 22q11.2 Microdeletion Syndrome Using Prequel® Prenatal Screen
SALT LAKE CITY, April 16, 2024 (GLOBE NEWSWIRE) -- Myriad Genetics, Inc., (NASDAQ: MYGN), a leader in genetic testing and precision medicine, today announced that Prenatal Diagnosis has published a study1 demonstrating exceptional positive predictive value (PPV) for 22q11.2 microdeletion screening using Myriad’s prenatal cell-free DNA (pcfDNA) screen, Prequel®, which incorporates fetal fraction amplification. Prior studies from other commercial pcfDNA laboratories have reported a broad range of
 Zacks
ZacksMyriad Genetics (MYGN) Announces Favorable Research Results
Myriad Genetics' (MYGN) latest positive initial study outcome is likely to improve clinical outcomes and reduce overall healthcare costs.