Ionis (IONS) Q3 Earnings & Revenues Fall Short of Estimates
Ionis Pharmaceuticals IONS reported third-quarter 2021 loss per share of 58 cents, which was wider than the Zacks Consensus Estimate of a loss of 53 cents. In the year-ago period, the company had incurred a loss of 18 cents per share.
The bottom line includes expenses related to the Akcea acquisition and restructured European and North American operations and other items. Excluding these non-recurring expenses, the loss per share was 34 cents per share. In the year-ago quarter, it recorded earnings of 9 cents per share.
In October 2020, Ionis acquired the remaining stake in Akcea Therapeutics and the latter became its wholly-owned subsidiary. In the first half of 2021, Ionis completed the transition of the commercial operations of its two commercialized products, Tegsedi (hereditary TTR amyloidosis, or hATTR) and Waylivra (genetically confirmed familial chylomicronemia syndrome, or FCS), in Europe and North America to Swedish Orphan Biovitrum AB, or Sobi, per a distribution agreement with the company. With these deals, its European and North American operations for Tegsedi and Waylivra were restructured from direct sales to a distribution model.
Ionis reported total revenues of $133 million, down 16.9% year over year due to lower R&D as well as commercial revenues. Sales missed the Zacks Consensus Estimate of $160 million.
This year so far, Ionis’ shares have declined 40.3% compared with the industry’s decrease of 14.2%.

Image Source: Zacks Investment Research
Quarter in Detail
Ionis has licensed Spinraza to Biogen BIIB. Biogen is responsible for commercializing Spinraza, approved for treating spinal muscular atrophy, or SMA, worldwide. Ionis receives royalties from Biogen on Spinraza’s sales.
Ionis earns commercial revenues, primarily royalty payments on net sales of Spinraza and R&D revenues, from partnered medicines.
Commercial revenues were $85 million in the third quarter, down 10.5% year over year.
Commercial revenues from Spinraza royalties were $67 million, down 9.5% year over. Revenues from Tegsedi and Waylivra from distribution fees were $15 million compared with $19 million in the year-ago quarter. License and royalty revenues were $3 million in the quarter compared with $2 million in the year-ago quarter.
R&D revenues of $48 million declined 26.2% from the year-ago quarter. Ionis expects R&D revenues to be higher in the fourth quarter than the third as its partnered programs are advancing.
Adjusted operating costs rose 22.5% year over year to $185 million in the third quarter mainly driven by higher R&D costs as the company rapidly advances its wholly-owned late-stage pipeline. The SG&A expenses decreased in the quarter due to cost efficiencies realized from integrating Akcea and restructuring commercial operations.
Pipeline Update
Ionis has seven pivotal phase III studies ongoing for medicines (internal as well as partnered), which include tofersen for SOD1-ALS (amyotrophic lateral sclerosis); pelacarsen for cardiovascular disease due to elevated Lp(a) levels; olezarsen (formerly APOCIII-LRx) for FCS and severe hypertriglyceridemia, eplontersen for TTR amyloidosis and ION363 for amyotrophic lateral sclerosis, or ALS, with mutations in the fused in sarcoma gene, or FUS (FUS-ALS).
Biogen and Novartis NVS are its partners for tofersen and pelacarsen. Mixed data from the phase III VALOR study of tofersen in patients with SOD1-ALS was announced last month. In the study, the primary endpoint did not reach statistical significance. However, signs of reduced disease progression across multiple secondary and exploratory endpoints were observed. The phase III Lp(a) HORIZON study on pelacarsen reached 50% enrollment in the third quarter for which Ionis earned a $25 million milestone payment from Novartis.
Ionis is advancing and expanding its wholly-owned pipeline to drive future revenue growth and data from several of these programs is expected in 2021/2022. The acquisition of Akcea and the restructured EU operation for Tegsedi/Waylivra has strengthened Ionis’ financial position and allowed it to focus on progressing its pipeline.
Ionis is developing olezarsen for FCS, eplontersen for TTR and ION363 for FUS-ALS on its own in pivotal late-stage studies. It has completed enrollment in the phase III NEURO-TTRansform study of eplontersen in TTR polyneuropathy with data expected in mid-2022. Ionis is also developing eplontersen for the treatment of cardiomyopathy in the phase III CARDIO-TTRansform study. Ionis began a second phase III study of olezarsen for severe hypertriglyceridemia in November.
Ionis also has several other candidates in phase II development, which it is developing in partnership with Biogen, Roche RHHBY or on its own.
Ionis expects to have 12 or more products on the market in 2026 with over half from its wholly-owned pipeline.
2021 Guidance
Ionis maintained its 2021 total revenue guidance of more than $600 million. Its adjusted net loss is expected to be less than $100 million.
Adjusted operating expense is expected to be in the range of $710 million to $750 million. R&D costs are expected to increase in the fourth quarter as olezarsen and donidalorsen phase III studies get underway
Ionis currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Ionis Pharmaceuticals, Inc. Price, Consensus and EPS Surprise
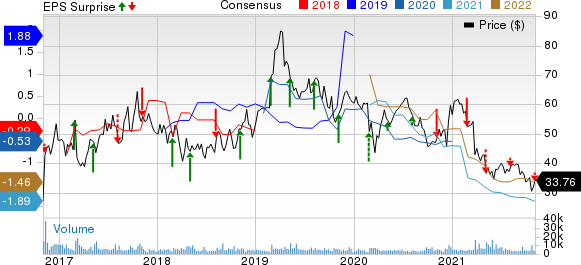
Ionis Pharmaceuticals, Inc. price-consensus-eps-surprise-chart | Ionis Pharmaceuticals, Inc. Quote
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Roche Holding AG (RHHBY) : Free Stock Analysis Report
Ionis Pharmaceuticals, Inc. (IONS) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 