Dynavax (DVAX) Up as EMA Accepts Valneva's COVID-19 Vaccine MAA
Shares of Dynavax Technologies Corporation DVAX gained 16.22% after partner Valneva SE VALN confirmed that the European Medicines Agency (“EMA”) has accepted the filing of a marketing authorization application (MAA) for its inactivated, whole-virus COVID-19 vaccine candidate, VLA2001.
VLA2001 consists of inactivated whole virus particles of SARS-CoV-2 with high S-protein density, in combination with two adjuvants, alum and CpG 1018. CpG 1018 adjuvant is supplied by Dynavax. It is also the component of
Dynavax’ FDA- and EMA-approved Heplisav vaccine. Hence, shares surged on the news.
The EMA acceptance implies the start of the formal review process by the EMA’s Committee for Human Medicinal Products (“CHMP”). If the committee accepts Valneva’s conditional MAA, the company expects to receive a positive CHMP opinion in June 2022.
Dynavax’s shares have lost 23% in the year so far compared with the industry’s decline of 24.1%.
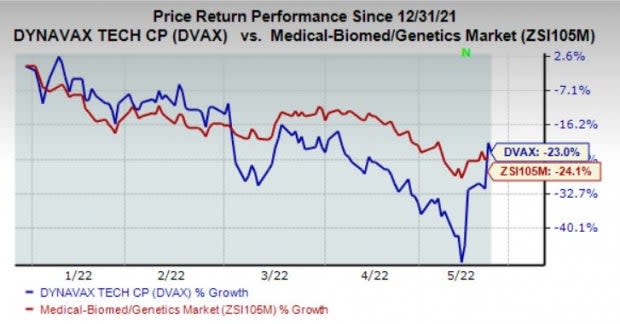
Image Source: Zacks Investment Research
Dynavax has established a global portfolio of CpG 1018 adjuvant commercial supply agreements currently focused on developing COVID-19 vaccines across a variety of vaccine platforms.
In April 2020, Dynavax entered into a collaboration agreement with Valneva to provide CpG 1018 adjuvant for use in the development of VALN’s COVID-19 vaccine candidate. The agreement was amended in July 2020 to provide additional quantities of CpG 1018 adjuvant. In September 2020, Dynavax entered into a supply agreement with Valneva to manufacture and supply specified quantities of CpG 1018 adjuvant for use in the commercialization of VLA2001.
CpG 1018 adjuvant revenues for the first quarter of 2022 were $91.5 million, up 23% from $74.6 million in the first quarter of 2021.
Dynavax expects full-year 2022 CpG 1018 adjuvant COVID-19 supply revenues to be at least $550 million, based on committed adjuvant orders, with full-year gross margin anticipated to be approximately 50%. DVAX is also advancing a pipeline of differentiated product candidates that leverage its CpG 1018 adjuvant.
It currently has a Zacks Rank #3 (Hold). Some better-ranked stocks are Alkermes ALKS and Geron Corporation GERN, both carrying a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Loss estimates for ALKS for 2022 have narrowed to 3 cents from a loss of 14 cents in the past 60 days. Alkermes surpassed estimates in all of the trailing four quarters, the average surprise being 350.48%.
Loss estimates for GERN for 2022 have narrowed by 6 cents in the past 60 days. Geron surpassed estimates in three of the trailing four quarters, the average surprise being 1.07%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Dynavax Technologies Corporation (DVAX) : Free Stock Analysis Report
Geron Corporation (GERN) : Free Stock Analysis Report
Alkermes plc (ALKS) : Free Stock Analysis Report
Valneva SE Sponsored ADR (VALN) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 